Global Value Dossiers (GVDs)

Clear, Evidence-Driven Value Communication for Global Market Access
A global value dossier (GVD) consolidates the clinical, economic, and wider evidence that payers, clinicians, and internal teams need to understand the value of your therapy across markets. As a central, strategic document, it supports global pricing, reimbursement planning, affiliate alignment, and consistent value communication throughout the product lifecycle.
At Petauri Evidence, we develop GVDs that are both scientifically robust and engaging, ensuring affiliates, payer-facing teams, and global functions can rely on a single, coherent, evidence-based value narrative.
A Compelling, Payer-Relevant Value Narrative
A clear, evidence-based value proposition sits at the heart of an effective market access strategy. It should form the core of your value dossier, bringing together the clinical, economic, and humanistic evidence into a cohesive story.
We work with you to develop messages that are:
- Evidence driven
- Relevant to payer priorities
- Aligned with global strategy
- Adaptable for affiliate use
- Tested internally and externally as needed
This ensures your narrative is credible, differentiated, and applicable across a range of reimbursement environments.
Our interdisciplinary team supports every stage of narrative and dossier development, from shaping the value proposition, through evidence synthesis and economic modeling, to dossier creation, Joint Clinical Assessment (JCA)/health technology assessment (HTA) alignment, and digital enablement via our Interactive Global Value Platform.
What to include in your GVD:
We would recommend including:
- Value story and executive summary
- Disease burden and unmet need
- Product information and positioning
- Clinical evidence and safety profile
- Patient-reported and humanistic outcomes
- Economic value including cost-effectiveness and budget impact models
- Market access strategy and objection handling
- Supporting literature, data sources, and appendices
The best GVDs follow a structured, intuitive framework that provides global market access and local affiliate teams with the key messaging and evidence they need.
Presenting Your Value Narrative
We can present your global value story and supporting evidence in a variety of formats to share with key stakeholders and local affiliates.
Anything is possible; it all depends on the level of detail you wish to include. Our clients often commission a combination of the following options.
Global Value Dossier (GVD)
A GVD will include your full value narrative, along with a detailed overview of the medicine/device itself, the clinical evidence, and other relevant data. It is typically presented as an A4 Word document or PDF.
GVDs are ideal for sharing all the relevant information required by key stakeholders and local affiliates in key markets.
Global Reimbursement Dossier (GRD) / Reimbursement Support Dossier
A GRD or reimbursement support dossier provides comprehensive information typically required for HTA submissions. They are not country-specific but are designed to provide local submission teams with the information they would need to support reimbursement.
As such, in addition to the full value narrative, they will often include detailed systematic literature reviews and health economic models.
A reimbursement support dossier is ideal for:
- Detailing all the information and evidence needed to support HTA submissions in key markets
- Supporting early reimbursement routes like the EU JCA
Value Proposition Slide Deck
A global value proposition deck is often the most reader-friendly way of presenting your product narrative. It typically comprises an intuitive, engaging PowerPoint deck of 30–40 slides, outlining the key value story themes supported by key messages and data.
It will clearly convey the unmet need and differentiation narrative in a way that resonates with key payer archetypes, and will be professionally designed to ensure visual impact.
A Value Proposition deck is ideal for:
- Presenting a refined, impactful global value story that can be adapted depending on competitive environment and payer value drivers
- Providing visually engaging, centrally approved slides that can be selected or modified for use in local presentations
Objection Handler
An objection handler is a document that includes company-approved responses to questions and challenges that affiliates are likely to face from payers during access and reimbursement discussions.
Likely objections are identified in collaboration with the client team by interrogation of the product evidence and competitor landscape, and by testing the value narrative with payers and key opinion leaders (KOLs), if required.
The document should be easy to use and navigate at speed, with clear, evidence-based responses that resonate with payer decision drivers in key markets.
An objection handler is ideal for:
- Providing a repository of robust, company-approved responses to anticipated payer concerns
- Reinforcing the product value story, ensuring each response ties back to the overarching unmet need and differentiation narrative
- Empowering field teams to confidently engage in reimbursement discussions and negotiations
Additional supporting communication materials
We also provide a range of other tools and activities to support you in communicating your global product value story and evidence.
Some of these are listed below, but we are always happy to discuss creating something more bespoke if required.
- Value story user guides and scripts
- GVD summary slide decks
- Deep dive educational slide decks
- Evidence compendiums
- Print-ready leave pieces
- Roll-out training and workshops
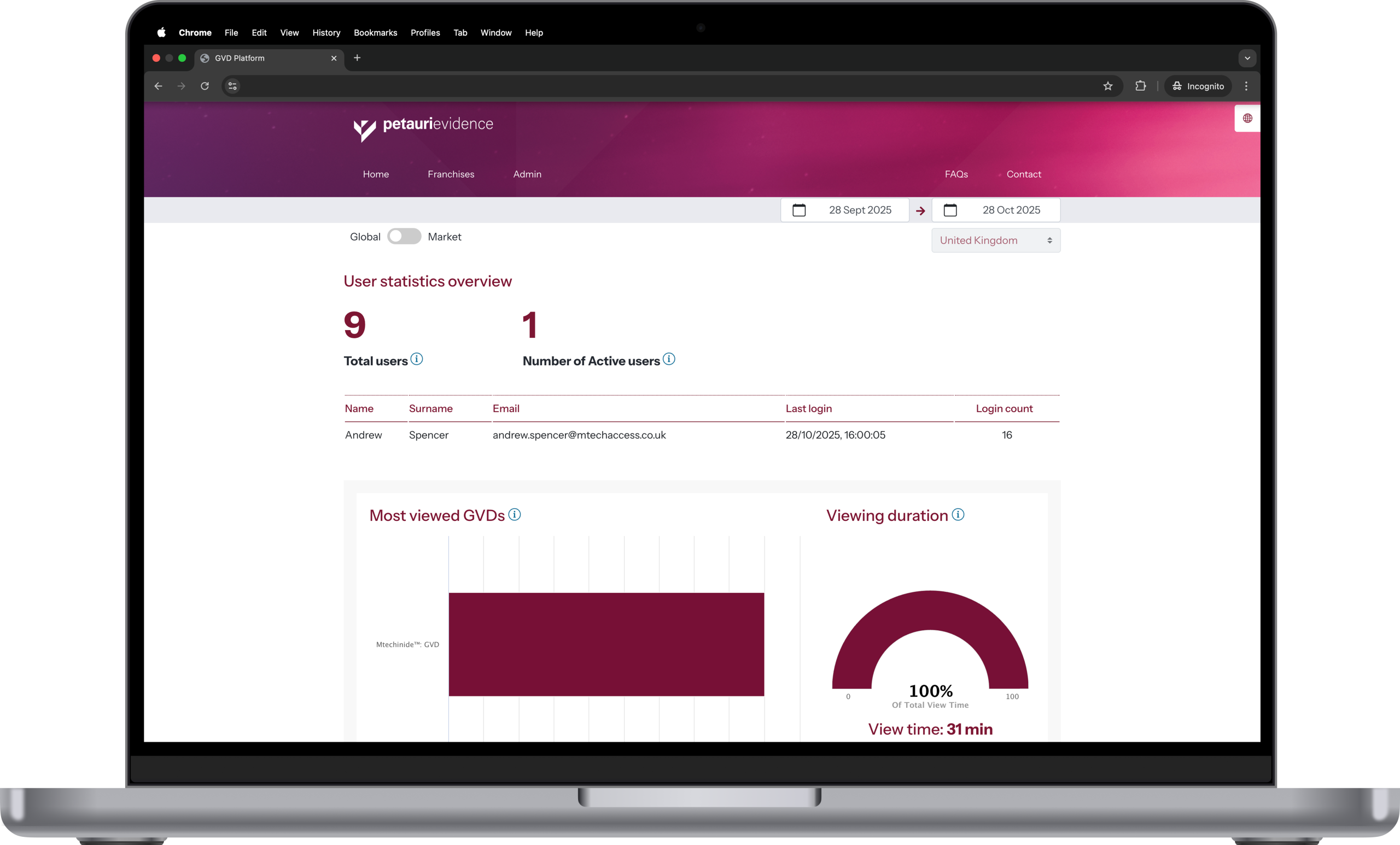
The Interactive Global Value Platform
Petauri’s Interactive Global Value Platform takes your global value communication materials far beyond static documents.
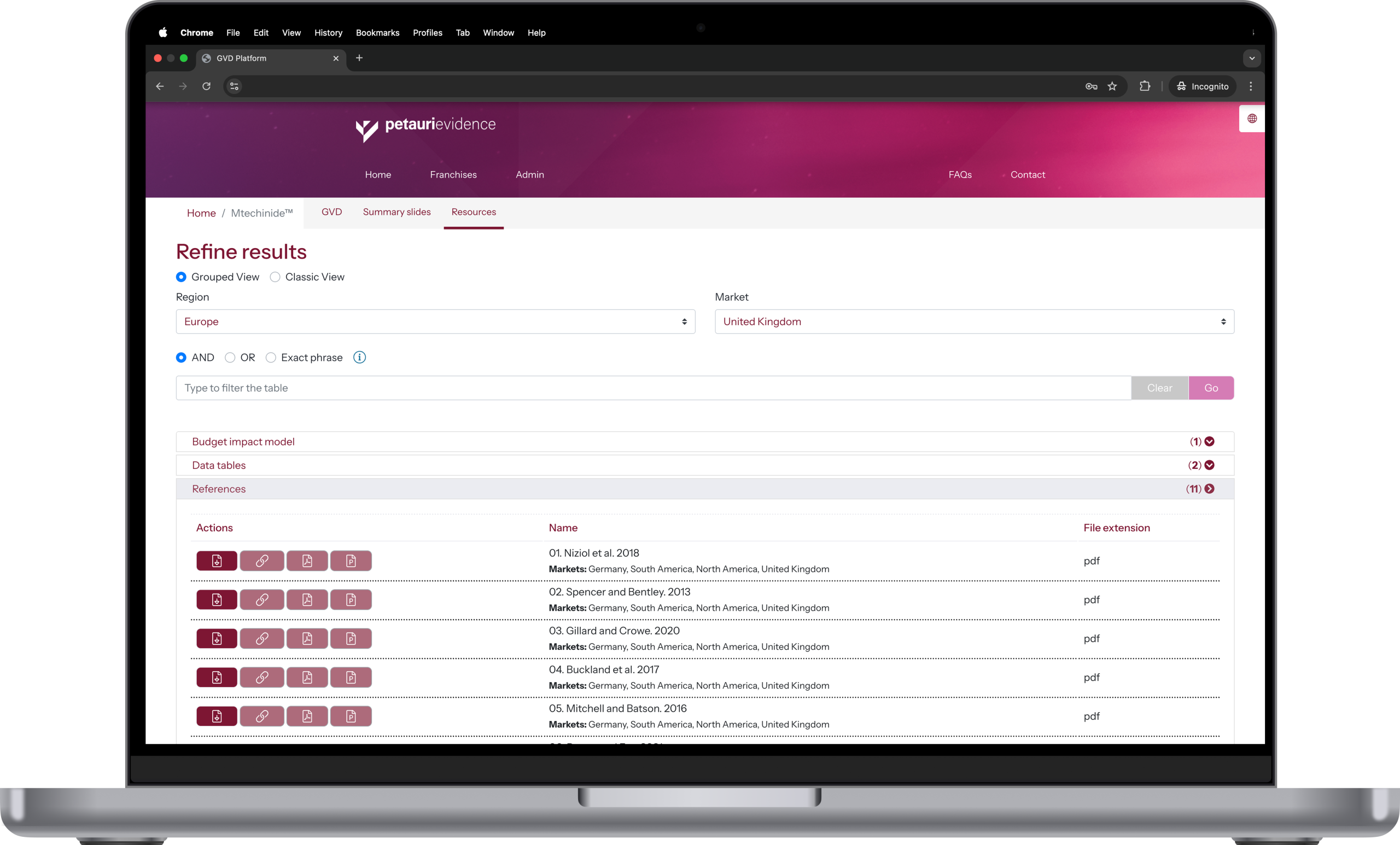
Our platform allows global teams to house:
- GVDs and reimbursement dossiers
- Summary decks and other supporting value communication materials
- Economic models and local variants
- Approved literature and evidence libraries
- Market specific content, tailored for affiliate access
Key benefits include:
- Always uptodate, centrally controlled content
- Easy navigation between dossier sections and supporting evidence
- Local affiliate tailoring without compromising global consistency
- Streamlined updates, version management, and crossmarket alignment
Global Evidence Dossiers Aligned with HTA and JCA Requirements
With evolving global expectations for evidence and clinical assessment, global evidence dossiers must be developed with future HTA needs in mind.
This includes emerging requirements under the EU JCA, where early alignment on comparators, endpoints, and PICOs scenarios can influence how global dossiers are used downstream.
Learn more about our JCA services- Anticipates likely HTA and JCA evidence expectations
- Uses structures and narratives that can be adapted into regional and national submissions
- Is built to support JCA ready clinical and economic content
- Provides a strong foundation for efficient development of JCA dossiers and national HTA submissions
Partner With Petauri
If you’re looking to develop a new Global Value Dossier, refresh an existing one, or modernize your approach with an interactive platform, our team can help.
Get in touch to discuss your project.
